Electrochemical Biosensors for Real-Time Oxidative Stress Monitoring in Saliva
DOI:
https://doi.org/10.64060/jestt.v2i2.3Keywords:
Slaviary biomarkers ofoxidative stress , Electrochemcal Biosensors, wearable sensingAbstract
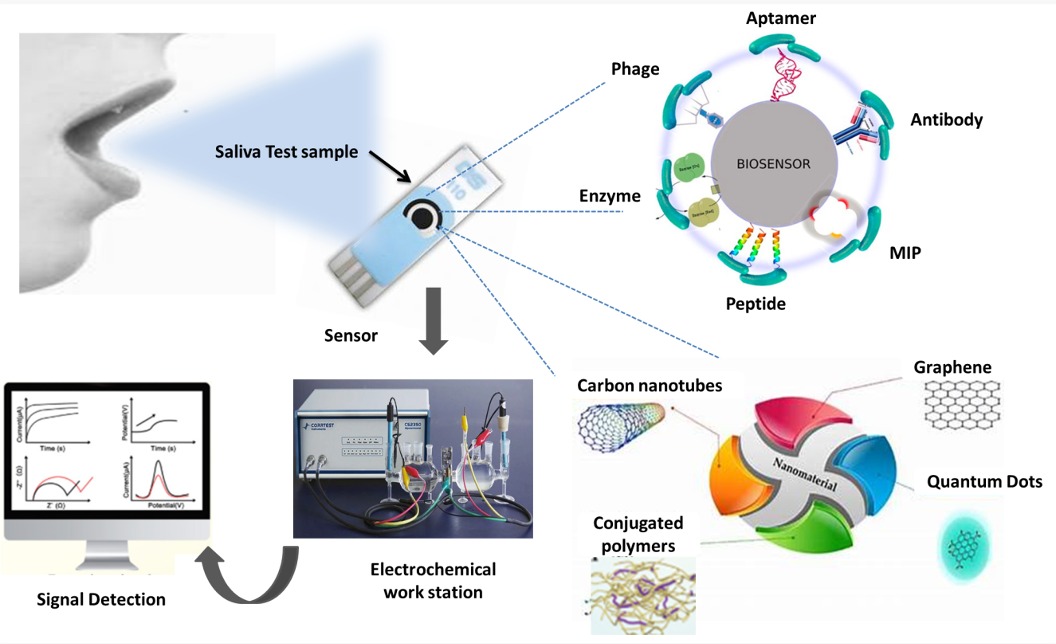
Oxidative stress, resulting from an imbalance between reactive oxygen species and antioxidant defences, is a critical factor in the onset and progression of numerous diseases, including cancer, cardiovascular conditions, and neurological disorders. Early detection of oxidative stress biomarkers is essential for timely diagnosis and effective treatment. Saliva has emerged as a highly attractive biofluid for this purpose due to its non-invasive, easily accessible, and cost-effective collection. Recent advancements in electrochemical biosensors have significantly enhanced the sensitivity, selectivity, and reliability of detecting oxidative stress indicators in saliva. These innovative sensing platforms enable real-time monitoring of key biomarkers at low concentrations, offering great potential for clinical and point-of-care applications. However, challenges such as sensor stability, biofouling, and interference from complex salivary components remain to be addressed to ensure robust performance in practical settings. This review summarises the latest developments in electrochemical biosensing of salivary oxidative stress biomarkers, highlights existing limitations, and discusses prospective strategies to overcome current barriers. The continued evolution of this technology promises to facilitate early disease detection and improve patient outcomes through accessible and precise oxidative stress monitoring.
References
1. S. Deshpande, W. Muraoka, and S. Andreescu, “Electrochemical sen-sors for oxidative stress monitoring,” Curr. Opin. Electrochem., vol. 29, p. 100809, Oct. 2021, doi: 10.1016/j.coelec.2021.100809.
2. M. P. Murphy et al., “Unraveling the Biological Roles of Reactive Oxy-gen Species,” Cell Metab., vol. 13, no. 4, pp. 361–366, Apr. 2011, doi: 10.1016/j.cmet.2011.03.010.
3. M. Schrader and H. D. Fahimi, “Peroxisomes and oxidative stress,” Bio-chim. Biophys. Acta BBA - Mol. Cell Res., vol. 1763, no. 12, pp. 1755–1766, Dec. 2006, doi: 10.1016/j.bbamcr.2006.09.006.
4. K. M. Nash, A. Rockenbauer, and F. A. Villamena, “Reactive Nitrogen Species Reactivities with Nitrones: Theoretical and Experimental Stud-ies,” Chem. Res. Toxicol., vol. 25, no. 8, pp. 1581–1597, Aug. 2012, doi: 10.1021/tx200526y.
5. H. Sies and D. P. Jones, “Reactive oxygen species (ROS) as pleiotropic physiological signalling agents,” Nat. Rev. Mol. Cell Biol., vol. 21, no. 7, pp. 363–383, Jul. 2020, doi: 10.1038/s41580-020-0230-3.
6. Y. Chen, M. B. Azad, and S. B. Gibson, “Superoxide is the major reac-tive oxygen species regulating autophagy,” Cell Death Differ., vol. 16, no. 7, pp. 1040–1052, Jul. 2009, doi: 10.1038/cdd.2009.49.
7. A. Andries, J. Rozenski, P. Vermeersch, D. Mekahli, and A. Van Schep-dael, “Recent progress in the LC–MS/MS analysis of oxidative stress bi-omarkers,” ELECTROPHORESIS, vol. 42, no. 4, pp. 402–428, 2021, doi: 10.1002/elps.202000208.
8. H. Togashi, M. Aoyama, and K. Oikawa, “Imaging of reactive oxygen species generated in vivo,” Magn. Reson. Med., vol. 75, no. 3, pp. 1375–1379, 2016, doi: 10.1002/mrm.25582.
9. M. A. Javaid, A. S. Ahmed, R. Durand, and S. D. Tran, “Saliva as a diagnostic tool for oral and systemic diseases,” J. Oral Biol. Craniofacial Res., vol. 6, no. 1, pp. 67–76, Jan. 2016, doi: 10.1016/j.jobcr.2015.08.006.
10. W. V. Giannobile, T. Beikler, J. S. Kinney, C. A. Ramseier, T. Morelli, and D. T. Wong, “Saliva as a diagnostic tool for periodontal disease: cur-rent state and future directions,” Periodontol. 2000, vol. 50, pp. 52–64, 2009, doi: 10.1111/j.1600-0757.2008.00288.x.
11. R. Monošík, M. Stred’anský, and E. Šturdík, “Application of Electro-chemical Biosensors in Clinical Diagnosis,” J. Clin. Lab. Anal., vol. 26, no. 1, pp. 22–34, 2012, doi: 10.1002/jcla.20500.
12. M. U. Javed et al., “Metal-Organic Framework/Polymer Composites for Solid State Electrolytes-A Critical Review,” J. Power Sources, vol. 640, p. 236720, Jun. 2025, doi: 10.1016/j.jpowsour.2025.236720.
13. F. J. Iftikhar, A. Shah, Q. Wali, and T. Kokab, “Advancements in Nano-fiber-Based Electrochemical Biosensors for Diagnostic Applications,” Biosensors, vol. 13, no. 4, Art. no. 4, Apr. 2023, doi: 10.3390/bios13040416.
14. Y. Li, “Molecularly imprinted polymer-based electrochemical biosensors for biomarkers detection,” Doctoral, UCL (University College London), 2024. Accessed: May 17, 2025. [Online]. Available: https://discovery.ucl.ac.uk/id/eprint/10195540/
15. I. D. Mandel, “Sialochemistry in Diseases and Clinical Situations Affect-ing Salivary Glands,” CRC Crit. Rev. Clin. Lab. Sci., vol. 12, no. 4, pp. 321–366, Jan. 1980, doi: 10.3109/10408368009108733.
16. U.-M. Bailey, C. Punyadeera, J. J. Cooper-White, and B. L. Schulz, “Analysis of the extreme diversity of salivary alpha-amylase isoforms generated by physiological proteolysis using liquid chromatography–tandem mass spectrometry,” J. Chromatogr. B, vol. 911, pp. 21–26, Dec. 2012, doi: 10.1016/j.jchromb.2012.10.023.
17. Y. Kanamaru, Kikukawa ,Azusa, and K. and Shimamura, “Salivary chromogranin-A as a marker of psychological stress during a cognitive test battery in humans,” Stress, vol. 9, no. 3, pp. 127–131, Jan. 2006, doi: 10.1080/14769670600909594.
18. F. P. Idris, Y. Wan, X. Zhang, and C. Punyadeera, “Within-Day Baseline Variation in Salivary Biomarkers in Healthy Men,” OMICS J. Integr. Bi-ol., vol. 21, no. 2, pp. 74–80, Feb. 2017, doi: 10.1089/omi.2016.0168.
19. M. A. D’amico, B. Ghinassi, P. Izzicupo, L. Manzoli, and A. D. Baldas-sarre, “Biological function and clinical relevance of chromogranin A and derived peptides,” Jun. 2014, doi: 10.1530/EC-14-0027.
20. A. L. Mandel, Ozdener ,Hakan, and V. and Utermohlen, “Brain-Derived Neurotrophic Factor in Human Saliva: Elisa Optimization and Biological Correlates,” J. Immunoassay Immunochem., vol. 32, no. 1, pp. 18–30, Jan. 2011, doi: 10.1080/15321819.2011.538625.
21. D. Tsikas, “Analytical methods for 3-nitrotyrosine quantification in bio-logical samples: the unique role of tandem mass spectrometry,” Amino Acids, vol. 42, no. 1, pp. 45–63, Jan. 2012, doi: 10.1007/s00726-010-0604-5.
22. S. Maheshwaran et al., “An Ultra-sensitive Electrochemical Sensor for the Detection of Oxidative Stress Biomarker 3-Nitro-l-tyrosine in Human Blood Serum and Saliva Samples Based on Reduced Graphene Oxide Entrapped Zirconium (IV) Oxide,” J. Electrochem. Soc., vol. 167, no. 6, p. 066517, Apr. 2020, doi: 10.1149/1945-7111/ab847d.
23. M. Hasanzadeh, F. Mokhtari, V. Jouyban-Gharamaleki, A. Mokhtarza-deh, and N. Shadjou, “Electrochemical monitoring of malondialdehyde biomarker in biological samples via electropolymerized amino ac-id/chitosan nanocomposite,” J. Mol. Recognit., vol. 31, no. 8, p. e2717, 2018, doi: 10.1002/jmr.2717.
24. J. Wang, H. M. Schipper, A. M. Velly, S. Mohit, and M. Gornitsky, “Sali-vary biomarkers of oxidative stress: A critical review,” Free Radic. Biol. Med., vol. 85, pp. 95–104, Aug. 2015, doi: 10.1016/j.freeradbiomed.2015.04.005.
25. H. A. Saputra, “Electrochemical sensors: basic principles, engineering, and state of the art,” Monatshefte Für Chem. - Chem. Mon., vol. 154, no. 10, pp. 1083–1100, Oct. 2023, doi: 10.1007/s00706-023-03113-z.
26. D. Grieshaber, R. MacKenzie, J. Vörös, and E. Reimhult, “Electrochem-ical Biosensors - Sensor Principles and Architectures,” Sensors, vol. 8, no. 3, Art. no. 3, Mar. 2008, doi: 10.3390/s80314000.
27. J. Baranwal, B. Barse, G. Gatto, G. Broncova, and A. Kumar, “Electro-chemical Sensors and Their Applications: A Review,” Chemosensors, vol. 10, no. 9, Art. no. 9, Sep. 2022, doi: 10.3390/chemosensors10090363.
28. A. M. Oliveira-Brett, J. A. P. Piedade, L. A. Silva, and V. C. Diculescu, “Voltammetric determination of all DNA nucleotides,” Anal. Biochem., vol. 332, no. 2, pp. 321–329, Sep. 2004, doi: 10.1016/j.ab.2004.06.021.
29. R. N. Goyal and G. Dryhurst, “Redox chemistry of guanine and 8-oxyguanine and a comparison of the peroxidase-catalyzed and electro-chemical oxidation of 8-oxyguanine,” J. Electroanal. Chem. Interfacial Electrochem., vol. 135, no. 1, pp. 75–91, Apr. 1982, doi: 10.1016/0022-0728(82)90006-7.
30. T.-H. Li, W.-L. Jia, H.-S. Wang, and R.-M. Liu, “Electrochemical per-formance of 8-hydroxy-2′-deoxyguanosine and its detection at poly(3-methylthiophene) modified glassy carbon electrode,” Biosens. Bioelec-tron., vol. 22, no. 7, pp. 1245–1250, Feb. 2007, doi: 10.1016/j.bios.2006.05.005.
31. X. Sun et al., “Electrochemical detection of 8-hydroxy-2′-deoxyguanosine as a biomarker for oxidative DNA damage in HEK293 cells exposed to 3-chloro-1,2-propanediol,” Anal. Methods, vol. 7, no. 16, pp. 6664–6671, Aug. 2015, doi: 10.1039/C5AY01246E.
32. A. Mugweru, B. Wang, and J. Rusling, “Voltammetric Sensor for Oxi-dized DNA Using Ultrathin Films of Osmium and Ruthenium Metallopol-ymers,” Anal. Chem., vol. 76, no. 18, pp. 5557–5563, Sep. 2004, doi: 10.1021/ac049375j.
33. N. Kumar, Rosy, and R. N. Goyal, “A melamine based molecularly im-printed sensor for the determination of 8-hydroxydeoxyguanosine in hu-man urine,” Talanta, vol. 166, pp. 215–222, May 2017, doi: 10.1016/j.talanta.2017.01.058.
34. G. V. Martins, A. C. Marques, E. Fortunato, and M. G. F. Sales, “8-hydroxy-2′-deoxyguanosine (8-OHdG) biomarker detection down to picoMolar level on a plastic antibody film,” Biosens. Bioelectron., vol. 86, pp. 225–234, Dec. 2016, doi: 10.1016/j.bios.2016.06.052.
35. A. M. Faria, E. B. M. I. Peixoto, C. B. Adamo, A. Flacker, E. Longo, and T. Mazon, “Controlling parameters and characteristics of electrochemi-cal biosensors for enhanced detection of 8-hydroxy-2′-deoxyguanosine,” Sci. Rep., vol. 9, no. 1, p. 7411, May 2019, doi: 10.1038/s41598-019-43680-y.
36. L.-P. Jia et al., “Enzyme-free and triple-amplified electrochemical sens-ing of 8-hydroxy-2′-deoxyguanosine by three kinds of short pDNA-driven catalyzed hairpin assemblies followed by a hybridization chain reaction,” Analyst, vol. 145, no. 10, pp. 3605–3611, May 2020, doi: 10.1039/D0AN00233J.
37. L. Yuan, Y. Lan, M. Han, J. Bao, W. Tu, and Z. Dai, “Label-free and facile electrochemical biosensing using carbon nanotubes for malondial-dehyde detection,” Analyst, vol. 138, no. 11, pp. 3131–3134, May 2013, doi: 10.1039/C3AN00041A
38. M. Jamshidi, A. Walcarius, M. Thangamuthu, M. Mehrgardi, and A. Ranjbar, “Electrochemical approaches based on micro- and nanomateri-als for diagnosing oxidative stress,” Microchim. Acta, vol. 190, no. 4, p. 117, Mar. 2023, doi: 10.1007/s00604-023-05681-7.
39. C. Amatore, S. Arbault, M. Guille, and F. Lemaître, “Electrochemical Monitoring of Single Cell Secretion: Vesicular Exocytosis and Oxidative Stress,” Chem. Rev., vol. 108, no. 7, pp. 2585–2621, Jul. 2008, doi: 10.1021/cr068062g.
40. M. K. Beissenhirtz, F. W. Scheller, and F. Lisdat, “A Superoxide Sensor Based on a Multilayer Cytochrome c Electrode,” Anal. Chem., vol. 76, no. 16, pp. 4665–4671, Aug. 2004, doi: 10.1021/ac049738f.
41. Z. Wang, H. Zhao, Q. Gao, K. Chen, and M. Lan, “Facile synthesis of ultrathin two-dimensional graphene-like CeO2–TiO2 mesoporous nanosheet loaded with Ag nanoparticles for non-enzymatic electrochem-ical detection of superoxide anions in HepG2 cells,” Biosens. Bioelec-tron., vol. 184, p. 113236, Jul. 2021, doi: 10.1016/j.bios.2021.113236.
42. S. Zhao, Z. Shi, C. X. Guo, and C. M. Li, “A high-energy-state biomimet-ic enzyme of oxygen-deficient MnTiO3 nanodiscs for sensitive electro-chemical sensing of the superoxide anion,” Chem. Commun., vol. 55, no. 54, pp. 7836–7839, Jul. 2019, doi: 10.1039/C9CC02679G.
43. E. Dumitrescu, K. N. Wallace, and S. Andreescu, “Real time electro-chemical investigation of the release, distribution and modulation of nitric oxide in the intestine of individual zebrafish embryos,” Nitric Oxide, vol. 74, pp. 32–38, Apr. 2018, doi: 10.1016/j.niox.2018.01.002.
44. R. E. Özel, K. N. Wallace, and S. Andreescu, “Chitosan coated carbon fiber microelectrode for selective in vivo detection of neurotransmitters in live zebrafish embryos,” Anal. Chim. Acta, vol. 695, no. 1, pp. 89–95, Jun. 2011, doi: 10.1016/j.aca.2011.03.057.
45. G. Ferrer-Sueta and R. Radi, “Chemical Biology of Peroxynitrite: Kinet-ics, Diffusion, and Radicals,” ACS Chem. Biol., vol. 4, no. 3, pp. 161–177, Mar. 2009, doi: 10.1021/cb800279q.
46. M. Govindasamy, S. Manavalan, S.-M. Chen, R. Umamaheswari, and T.-W. Chen, “Determination of oxidative stress biomarker 3-nitro-l-tyrosine using CdWO4 nanodots decorated reduced graphene oxide,” Sens. Ac-tuators B Chem., vol. 272, pp. 274–281, Nov. 2018, doi: 10.1016/j.snb.2018.05.138.
47. R. Jalili and M. Amjadi, “Bio-inspired molecularly imprinted polymer–green emitting carbon dot composite for selective and sensitive detection of 3-nitrotyrosine as a biomarker,” Sens. Actuators B Chem., vol. 255, pp. 1072–1078, Feb. 2018, doi: 10.1016/j.snb.2017.08.145.
48. G. V. Martins, A. C. Marques, E. Fortunato, and M. G. F. Sales, “Wax-printed paper-based device for direct electrochemical detection of 3-nitrotyrosine,” Electrochimica Acta, vol. 284, pp. 60–68, Sep. 2018, doi: 10.1016/j.electacta.2018.07.150.
49. S.-M. Chen et al., “Hierarchically structured CuFe2O4 ND@RGO com-posite for the detection of oxidative stress biomarker in biological fluids,” Inorg. Chem. Front., vol. 5, no. 4, pp. 944–950, Apr. 2018, doi: 10.1039/C7QI00799J.
50. S. Wang et al., “Constructing a novel composite of molecularly imprinted polymer-coated AuNPs electrochemical sensor for the determination of 3-nitrotyrosine,” Electrochimica Acta, vol. 259, pp. 893–902, Jan. 2018, doi: 10.1016/j.electacta.2017.11.033.
51. S. P. Ng, G. Qiu, N. Ding, X. Lu, and C.-M. L. Wu, “Label-free detec-tion of 3-nitro-l-tyrosine with nickel-doped graphene localized surface plasmon resonance biosensor,” Biosens. Bioelectron., vol. 89, pp. 468–476, Mar. 2017, doi: 10.1016/j.bios.2016.04.017.
52. X. Wang, M. Zhu, H. Liu, J. Ma, and F. Li, “Modification of Pd–Fe na-noparticles for catalytic dechlorination of 2,4-dichlorophenol,” Sci. Total Environ., vol. 449, pp. 157–167, Apr. 2013, doi: 10.1016/j.scitotenv.2013.01.008.
53. Y. Bai and S. Li, “Oxidative Stress Sensing System for 8-OHdG Detec-tion Based on Plasma Coupled Electrochemistry by Transparent ITO/AuNTAs/PtNPs Electrode,” Biosensors, vol. 13, no. 6, p. 643, Jun. 2023, doi: 10.3390/bios13060643.
54. L. Yang, B. Wang, H. Qi, Q. Gao, C. Li, and C. Zhang, “Highly Sensitive Electrochemical Sensor for the Determination of 8-Hydroxy-2′-deoxyguanosine Incorporating SWCNTs-Nafion Composite Film,” J. Sens., vol. 2015, no. 1, p. 504869, 2015, doi: 10.1155/2015/504869.
55. Rosy and R. N. Goyal, “Determination of 8-Hydroxydeoxyguanosine: A potential biomarker of oxidative stress, using carbon-allotropic nano-materials modified glassy carbon sensor,” Talanta, vol. 161, pp. 735–742, Dec. 2016, doi: 10.1016/j.talanta.2016.09.038.
56. P. Montoro-Leal, M. Zougagh, A. Sánchez-Ruiz, Á. Ríos, and E. Vereda Alonso, “Magnetic graphene molecularly imprinted polypyrrole polymer (MGO@MIPy) for electrochemical sensing of malondialdehyde in serum samples,” Microchem. J., vol. 178, p. 107377, Jul. 2022, doi: 10.1016/j.microc.2022.107377.
57. B. Sriram, S. Kogularasu, S.-F. Wang, and G.-P. Chang-Chien, “The Fabrication of a La2Sn2O7/f-HNT Composite for Non-Enzymatic Elec-trochemical Detection of 3-Nitro-l-tyrosine in Biological Samples,” Bio-sensors, vol. 13, no. 7, Art. no. 7, Jul. 2023, doi: 10.3390/bios13070722.
58. B. Sriram, J. N. Baby, Y.-F. Hsu, S.-F. Wang, and M. George, “In Situ Synthesis of a Bismuth Vanadate/Molybdenum Disulfide Composite: An Electrochemical Tool for 3-Nitro-l-Tyrosine Analysis,” Inorg. Chem., vol. 61, no. 35, pp. 14046–14057, Sep. 2022, doi: 10.1021/acs.inorgchem.2c02037.
59. F. X. Hu et al., “Living Cells Directly Growing on a DNA/Mn3(PO4)2-Immobilized and Vertically Aligned CNT Array as a Free-Standing Hy-brid Film for Highly Sensitive In Situ Detection of Released Superoxide Anions,” Adv. Funct. Mater., vol. 25, no. 37, pp. 5924–5932, 2015, doi: 10.1002/adfm.201502341.
60. Z. Zhai et al., “Robust nanozyme with dense crystalline-amorphous inter-faces for efficient detection of reactive oxygen species in saliva,” Sens. Actuators B Chem., vol. 435, p. 137571, Jul. 2025, doi: 10.1016/j.snb.2025.137571.
61. U. Afzal et al., “Chemical engineering for advanced flexible sensors: Novel carbon-metal oxide nanocomposites with superior multi-sensing behavior,” Sens. Actuators B Chem., vol. 431, p. 137449, May 2025, doi: 10.1016/j.snb.2025.137449.
62. Z. Xu et al., “A wearable sensor based on multifunctional conductive hydrogel for simultaneous accurate pH and tyrosine monitoring in sweat,” Biosens. Bioelectron., vol. 234, p. 115360, Aug. 2023, doi: 10.1016/j.bios.2023.115360.
63. A. Khan, E. DeVoe, and S. Andreescu, “Carbon-based electrochemical biosensors as diagnostic platforms for connected decentralized healthcare,” May 2023, doi: 10.1039/D2SD00226D.
64. C. Mwanza, W.-Z. Zhang, K. Mulenga, and S.-N. Ding, “Advancing green chemistry in environmental monitoring: the role of electropolymer-ized molecularly imprinted polymer-based electrochemical sensors,” Green Chem., vol. 26, no. 23, pp. 11490–11517, Nov. 2024, doi: 10.1039/D4GC03250K.
65. J. Kim et al., “Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics,” Biosens. Bioelectron., vol. 74, pp. 1061–1068, Dec. 2015, doi: 10.1016/j.bios.2015.07.039.
66. H. Duan et al., “Wearable Electrochemical Biosensors for Advanced Healthcare Monitoring,” Adv. Sci. Weinh. Baden-Wurtt. Ger., vol. 12, no. 2, p. e2411433, Jan. 2025, doi: 10.1002/advs.202411433.
67. R. Diab, A. Choufani, J. Dagher, and N. Chahine, “The Influence of Circadian Rhythm on the Antioxidant Capacity of Saliva in Periodontal Diseases,” Cureus, vol. 16, no. 3, p. e56174, Mar. 2024, doi: 10.7759/cureus.56174.
68. S. Watanabe, Y. Kawasaki, and K. Kawai, “Diurnal variation of salivary oxidative stress marker 8-hydroxyguanine,” Genes Environ., vol. 41, no. 1, p. 20, Dec. 2019, doi: 10.1186/s41021-019-0138-3.
69. J. F. Hernández-Rodríguez, D. Rojas, and A. Escarpa, “Electrochemical Sensing Directions for Next-Generation Healthcare: Trends, Challenges, and Frontiers,” Anal. Chem., vol. 93, no. 1, pp. 167–183, Jan. 2021, doi: 10.1021/acs.analchem.0c04378.
70. P. V. V. Romanholo, C. A. Razzino, P. A. Raymundo-Pereira, T. M. Prado, S. A. S. Machado, and L. F. Sgobbi, “Biomimetic electrochemical sensors: New horizons and challenges in biosensing applications,” Bio-sens. Bioelectron., vol. 185, p. 113242, Aug. 2021, doi: 10.1016/j.bios.2021.113242.
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Journal of Engineering, Science and Technological Trends

This work is licensed under a Creative Commons Attribution 4.0 International License.